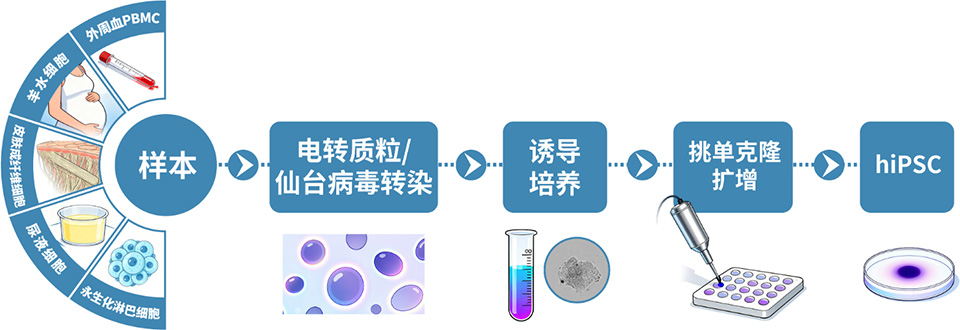
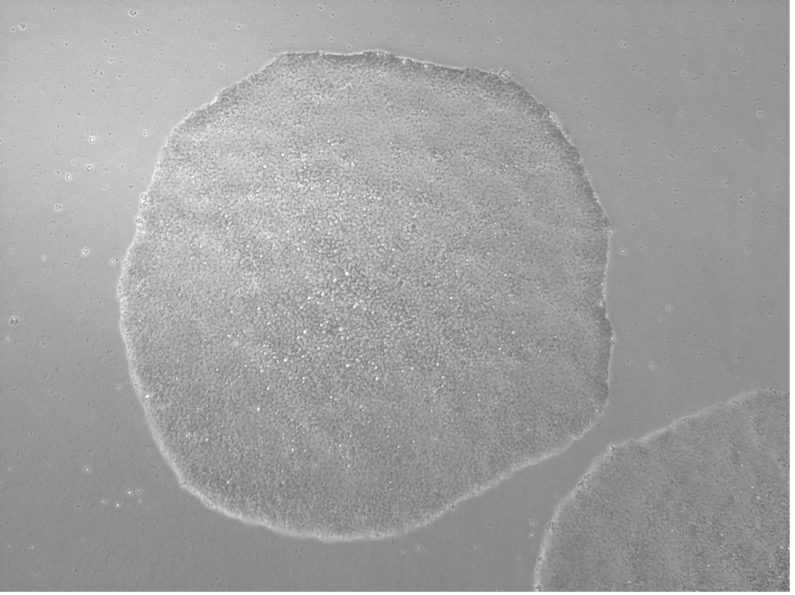
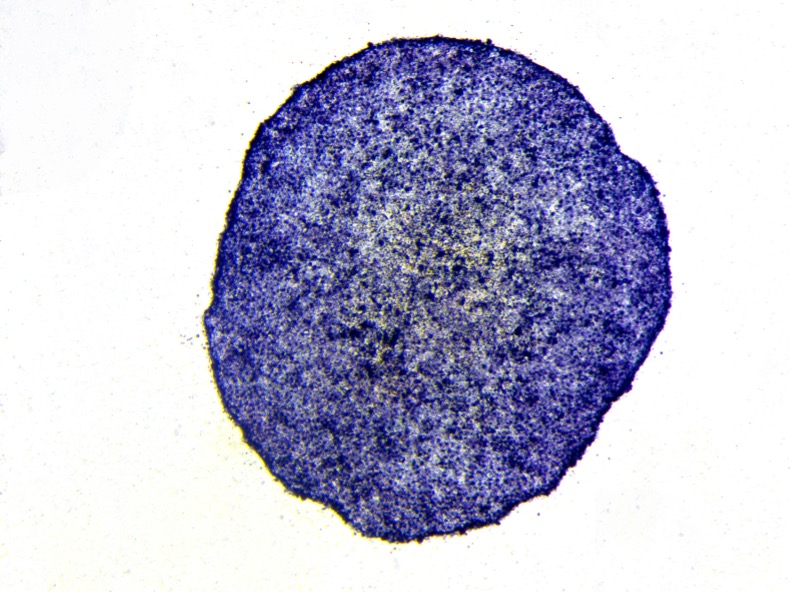
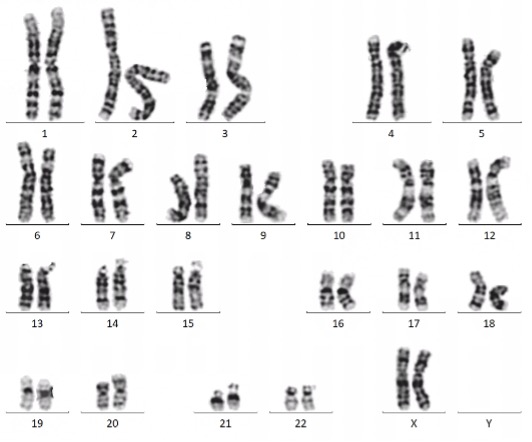
Wide range of sample sources for line generation
We are proficient in reprogramming technologies for a variety of somatic cells, and can generate cell lines from multiple sample sources including blood, amniotic fluid, skin, and urine, to meet your diverse project needs.
Mature Technology
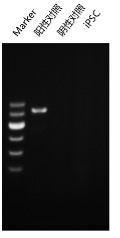
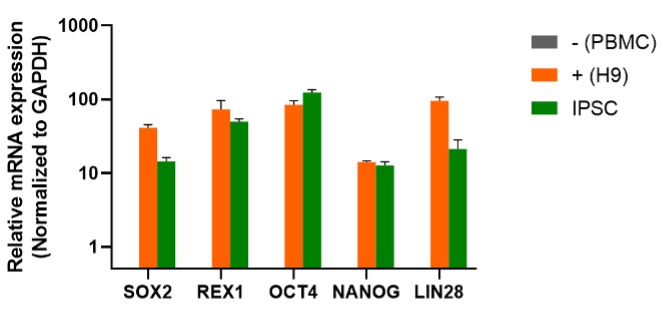
Our most well-established protocol is for iPSC line generation from peripheral blood mononuclear cells (PBMCs). The technical workflow has been iteratively optimized and validated, delivering industry-leading stability and efficiency.
Nationwide Accessible Biospecimen Collection Network
To ensure sample freshness and experimental success rate, we have professional PBMC isolation stations in East China, North China, South China, Northwest China and other regions, with a fully established, standardized workflow for nationwide biospecimen collection, isolation, and cold-chain transportation.
Strict Quality Control and Complete Deliverables
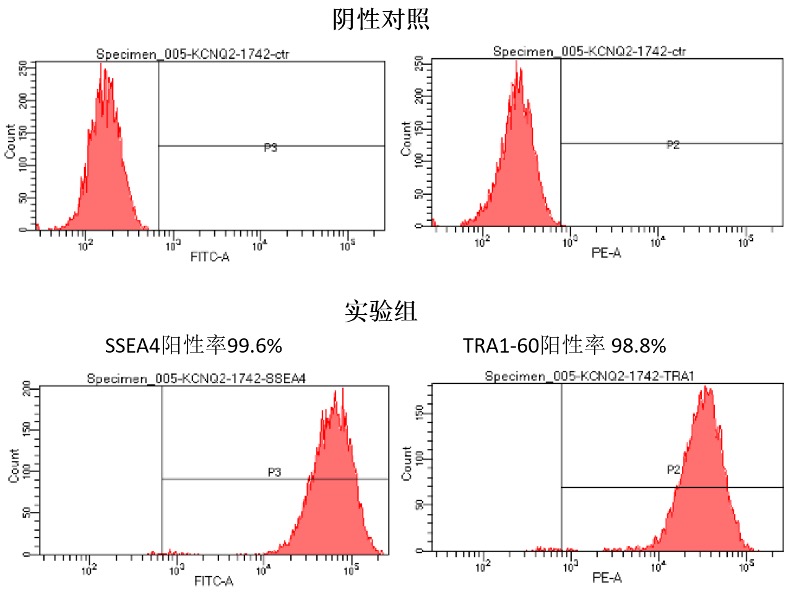
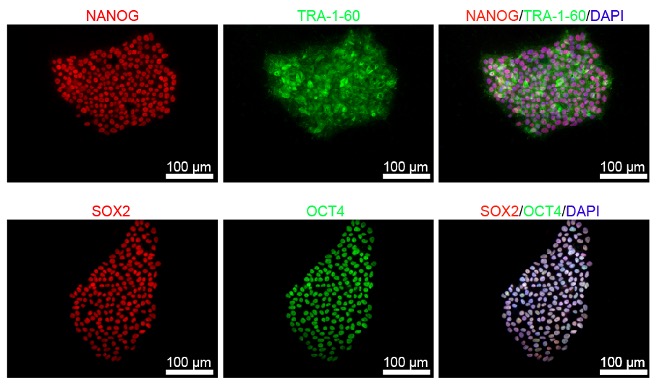
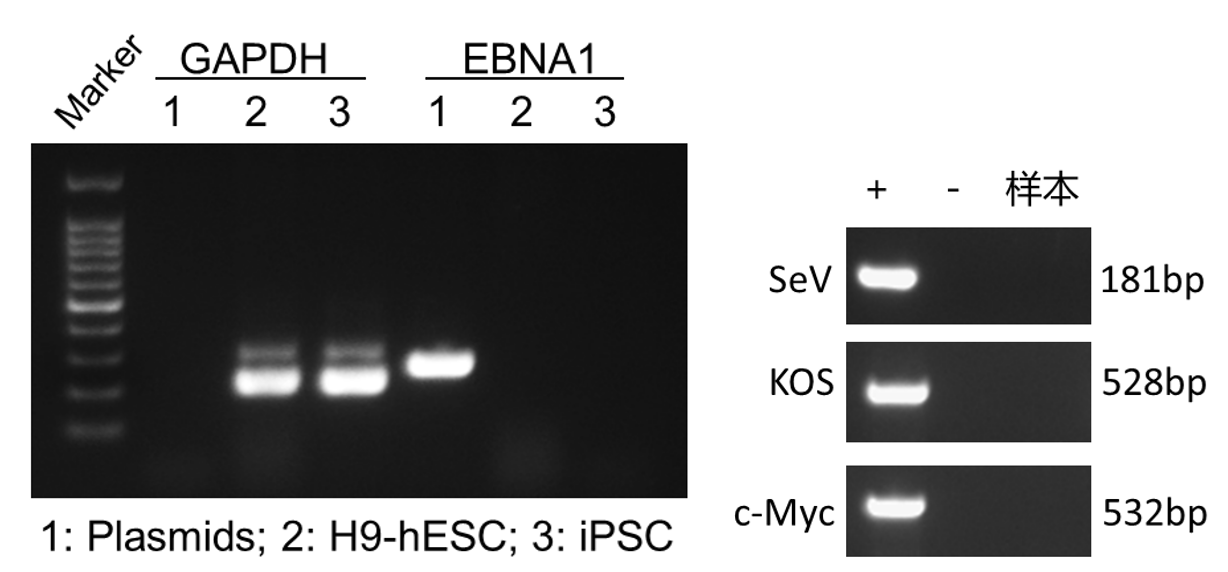
Upon successful line generation, we deliver 2 vials of monoclonal cell lines, accompanied by a comprehensive characterization report.