Primary microcephaly is a relatively rare disorder characterized by a significantly reduced head circumference. The main pathogenic cause is abnormal gene expression in neural stem cells, which impairs normal neurogenesis and leads to a reduced number of neurons. To date, 32 genes have been associated with microcephaly primary hereditary (MCPH), including MCPH1, ASPM, WDR62, among others. Most MCPH genes are involved in biological processes such as cell cycle regulation, centrosome assembly, DNA repair, and apoptosis.
Recently, the research groups of Bian Shan and Guo Zhenming from the School of Life Sciences and Technology, Tongji University, Wang Hua from Hunan Children's Hospital, and Mao Xiao from Hunan Maternal and Child Health Hospital clinically identified a microcephaly patient carrying a CETN3 deletion mutation. The patient's head circumference was 2–3 standard deviations below the normal range. However, the regulatory function of CETN3 in human cerebral cortex development has remained unclear. Thus, the researchers collaborated to explore the function of this gene in cortical development (Soma Biosciences participated in the construction of CETN3 mutant iPSCs). The findings were published in EMBO Molecular Medicine.
Centrin proteins belong to the EF‑hand superfamily of calcium‑binding proteins and contain four calcium‑binding domains. In cells, they are mainly localized in structures associated with microtubule‑organizing centers, such as centrosomes, cilia/flagella, and spindles. Most centrin proteins are also distributed in the cytoplasm and nucleus. Centrin proteins are highly conserved throughout evolution, but the number of genes encoding them varies among different organisms: mice have four (Cent1‑4), while humans have three (CETN1‑3). Their expression patterns differ across tissues and cell types, suggesting that distinct centrin proteins may participate in different biological processes.
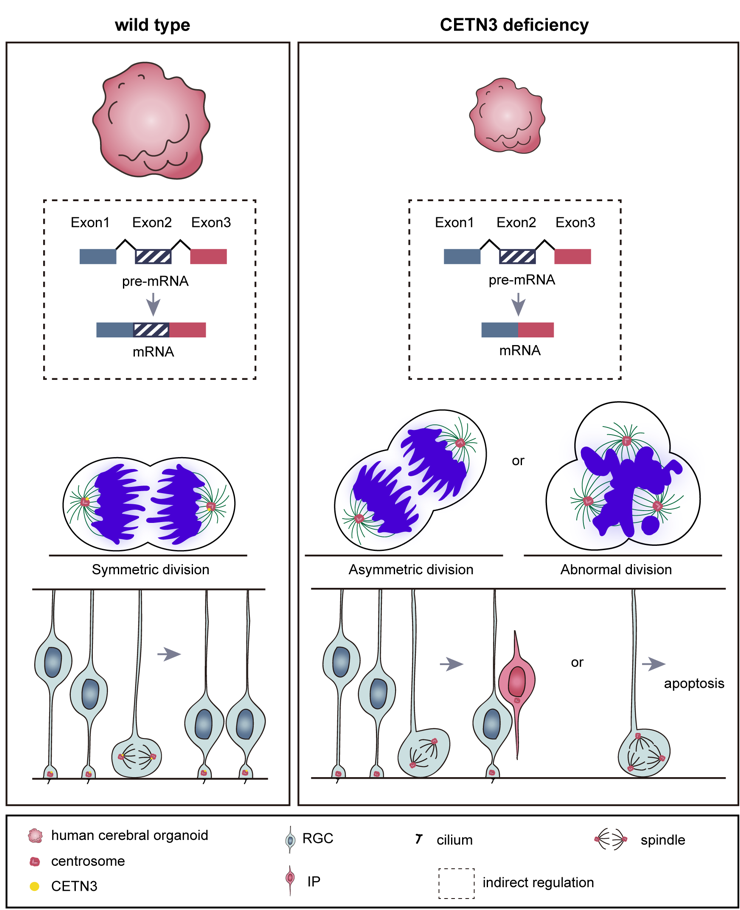
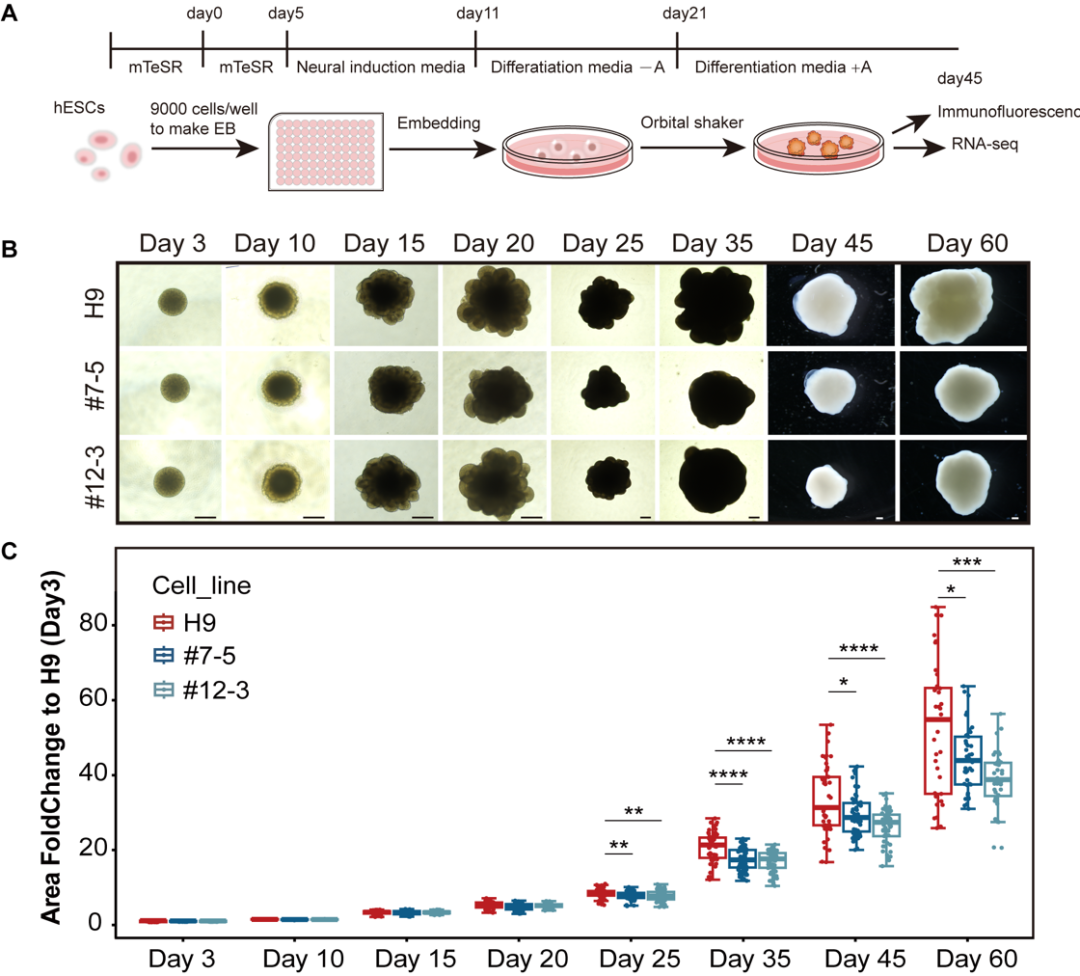
In this study, the authors focused on investigating the role of CETN3 in cerebral cortex development, assessing the pathogenic potential of CETN3 deletion mutations leading to microcephaly, and elucidating the underlying molecular mechanisms. First, they examined CETN3 expression in the cerebral cortex and found that it is broadly expressed in various cell types. Next, using CRISPR‑Cas9 and Cre‑LoxP gene‑editing technologies, they generated CETN3 knockout human embryonic stem cell (hESC) lines and CETN3 mutant induced pluripotent stem cell (iPSC) lines. Based on these cell lines, they obtained neural stem/progenitor cells (NS/PCs) through 2D culture and cerebral organoids through 3D culture. These organoid models recapitulated the reduced brain volume phenotype observed in patients, confirming the pathogenicity of CETN3.
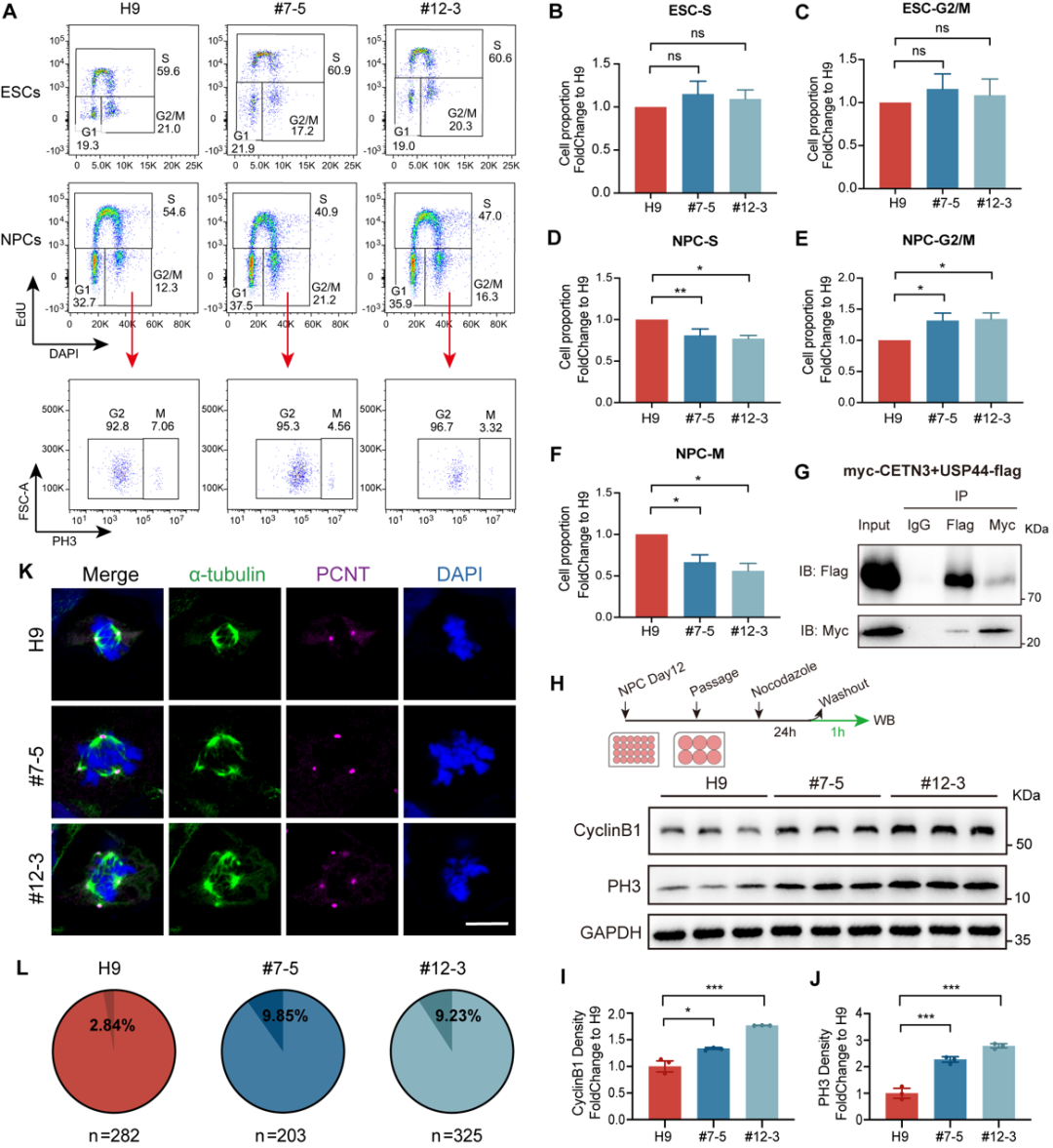
Next, using methods such as RNA‑seq, immunofluorescence staining, and flow cytometry, the authors further investigated the molecular mechanisms by which CETN3 regulates neural development. They found that CETN3 affects neurogenesis by modulating the proliferation, differentiation, apoptosis, and RNA splicing of neural stem/progenitor cells (NS/PCs), as detailed below:
(1) CETN3 deficiency alters the orientation of the mitotic spindle in NS/PCs, making them more prone to differentiation rather than proliferation;
(2) CETN3 deficiency impairs the NS/PC cell cycle, thereby affecting cell proliferation;
(3) CETN3 deficiency leads to centrosome overduplication, resulting in NS/PC apoptosis;
(4) CETN3 also indirectly affects the expression of genes related to centrioles, cilia, and the cell cycle by participating in RNA splicing.
Notably, this is the first report of a role for CETN3 in regulating RNA splicing. The authors also identified that this function of CETN3 may be associated with the deubiquitinating activity of USP49 and the ubiquitination level of histone H2B.
Finally, the authors also generated a forebrain‑specific CETN3 knockout mouse model to investigate whether CETN3 has similar functions in neural development in other mammals. The results showed only a mild phenotype in mice, suggesting that the function of CETN3 may differ across species.
In summary, using cerebral organoids as the primary model, this study reveals for the first time the role of CETN3 in mammalian neural development and the underlying molecular mechanisms. The research demonstrates that CETN3 participates in regulating the balance between neural progenitor cell (NPC) proliferation and differentiation by modulating biological processes such as spindle orientation, cell cycle progression, and centrosome duplication. The study also reports for the first time that CETN3 is involved in RNA splicing in NPCs through its interaction with USP49, thereby indirectly influencing the cell cycle and neural development. These findings highlight the importance of CETN3 in the development of the human neocortex, expand our understanding of causative genes in primary microcephaly, and provide new insights for future clinical diagnosis and therapeutic strategies.